探究影响合成多肽药物质量的常见因素与控制措施

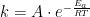
打开文本图片集
中图分类号: TQ207+ .1 文献标志码:A 文章编号:1001-5922(2026)03-0642-04
Investigation common factors and control measures affecting the quality of synthetic peptide drugs
ZHANG Yingya,ZHANG Xiting (CHIATAI Tianqing Pharmaceutical Group Co.,Ltd.,Lianyungang 222Ooo,Jiangsu China)
Abstract:The raw materials,solvents,reaction conditions and instruments,as wellas the purification process were investigated to identifythe key points affectingthequalityof synthetic polypeptides,and solid-phase peptide synthesis (SPPS)and liquid-phase peptide synthesis (LPPS)were discussd.In particular,the quality control of amino acids,the selection and dosage of solvents,and the control of the reaction process are important links to ensure the qualityof peptideproducts.Through thequality monitoring of products from diferent batches,high performance liquid chromatography-massspectrometry was adopted to control the purity,stability and biological activityof the products,which provided a reference basis for optimizing process conditions,establishing standard control methods and evaluating batch-to-batch consistency.
Key Words :synthetic peptide drugs;quality control;reagent reagents;batch consistency ;stability testing
随着多肽类药物广泛应用于生物医学领域,如何对其进行高质量生产过程监测就显得尤为重要。(剩余5887字)