糠醛脱羰制备呋喃的热力学及动力学分析

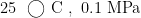
打开文本图片集
中图分类号:TQ013.1;TQ013.2 文献标志码:A 文章编号:1671-0460(2026)01-0211-04
Abstract:The research progress ofcatalysts for furfural decarboxylation to produce furan was summarized.The equilibrium reactor (REquil) and stoichiometric reactor (RStoic) modules in Aspen Plus software were employed to calculate the Gibbs free energy,reaction heat,and equilibrium composition ofthereaction.The effects of temperature, pressure,and feed composition on equilibrium conversion were systematically analyzed. Additionally, experimental kinetic data werecombined toestablishareaction kineticmodel,therateequation was derivedtoclarifytheratecontroling steps and identify key kinetic parameters (activation energy, pre-exponential factor),thereby revealing the regulatory mechanism of catalystsand reaction conditionson kinetic rates.Theresultsindicated that furfural decarboxylation was anendothermic reaction with an increase in the number ofgas molecules.At temperatureof 800~900K pressureof 0.05~0.10MPa ,and N2 to furfural molar ratio of 5~7, the equilibrium conversion exceeded 90% From a kinetic perspective,the reaction followed the Langmuir-Hinshelwood adsorption-kinetic model,wherethe adsorption of furfural on the catalyst active sites and the cleavage of the C—C bond were the rate-controllng steps. Catalysts such as PdC and Cr2O3/Al2O3 can reduce the activation energy to 60~85kJ⋅mol-1 , significantly enhancing the reaction rate.
Key words: Furfural; Decarboxylation reaction; Furan; Thermodynamic analysis; Kinetic model; Activation energy
呋喃作为重要的杂环化工中间体[1],广泛应用于医药、农药、高分子材料等领域[2-3]。(剩余6235字)