三羟甲基丙烷与羧酸的分步酯化反应动力学

打开文本图片集
中图分类号:TQ225.1 文献标志码:A 文章编号:1671-0460(2026)01-0014-07
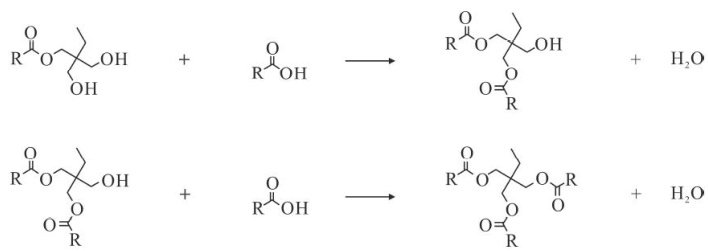
Abstract: The vacuum autocatalytic esterification process and reaction kinetics of trimethylolpropane with n-heptanoic acid,octanoic acid,and nonanoic acid were studied using online sampling method.The reaction kinetics parameters ofthestepwise esterification were obtained,andareaction kinetics model was established.The influence of carboxylic acid type and reaction temperature on esterification rate and product properties was analyzed.The results showed that the synthesis of trimethylolpropane ester by vacuum self-catalytic esterification method did not require catalyst, when the reaction temperature was 200~230∘C and the reaction time was 5h ,the esterification rate was over 90% ,and the mole fraction of triester reached 80%~97% .This reaction was an endothermic reaction, with activation energies ranging from 19kJ⋅mol-1 to 44kJ⋅mol-1 for each step. The calculated values from the established kinetic model were in good agreement with theexperimental values.The longer thealkyl chain,the more difficult the reaction,the lower the esterification rate,and the higher the mole fraction, kinematic viscosity,viscosityindex,and flash pointof the product's rimester.Raising thereaction temperature was beneficial for the formation oftriglycerides, and the temperature had little effect on the performance of product within a certain range.
Key Words: Trimethylolpropane; Heptanoic acid; Octanoic acid; Nonanoic acid; Esterification; Kinetics
润滑油在机械设备运行中起着不可或缺的作用,其市场需求量随着机械、交通运输等行业的迅速发展而逐年增加[1-2]。(剩余10275字)