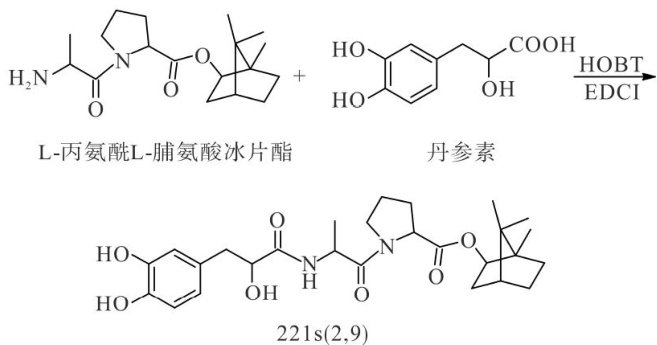
抗高血压1类新药—221s(2,9)的纯化工艺

打开文本图片集
中图分类号:TQ463;0621.3 文献标志码:A 文章编号:1671-0460(2026)01-0079-05
Abstract:Inordertoobtaintherapid purificationprocessconditions for theantihypertensivedrug221s(2,9),the crude product was preliminarily separated by using the pressured column chromatography method, the rapid column chromatography method with a sintered glassfunnel,and the continuous vacuum column chromatographymethod,and further purification operations were caried out in combination with the recrystallization method.The experimental results showed that when the sample-to-silica gel mass ratio is 1:10,the pressured column chromatography method had a goodseparation degreeandcould achieve acertain purificationefect whencombined with recrystallization.However, its efficiency and purification yield were relatively low. Although the rapid column chromatography method with a sintered glass funnel hadarelativelyhigheficiency,itsseparation effct was notsatisfactoryand the purificationgoal could not beachieved evenaftersubsequentsecondaryrecrystalization.When thesample tosilica gelmass ratio reached 1:12 in the continuous vacuum column chromatography method, the recovery rate could reach 83.5% and the crude separation purity was 93.2% , and the purification goal could be achieved through only one recrystallization. The purified method after optimization can rapidlyand eficiently purify221s(2,9),laying a solid foundation for the subsequent pilot-scale process research.
Keywords: 221s(2,9) ;Purification;Process; Optimization;Separation;Pharmaceuticals; Crystallization
随着人口结构的变化以及生活习惯的改变,高血压患病率日益增高,高血压给众多患者的健康造成了严重威胁,逐渐演变成了公共卫生问题[1-3]。(剩余7790字)