基于《中国医疗机构药品评价与遴选快速指南(第二版)》的静脉铁剂多维度量化评价

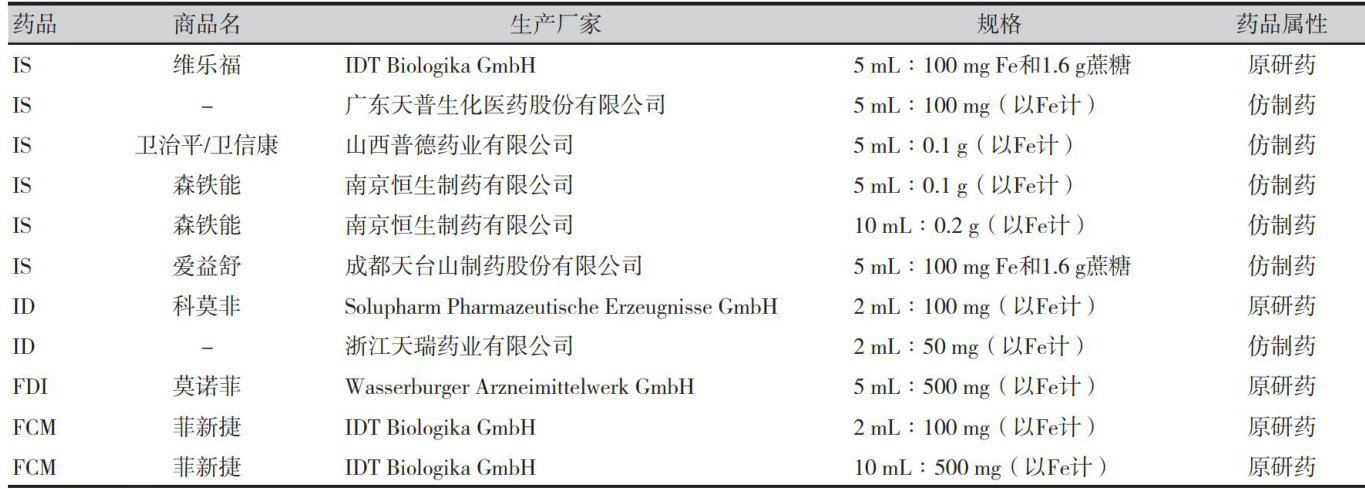
打开文本图片集
【中图分类号】R977 【文献标识码】A
【Abstract】 Objective To conduct a multi-dimensional quantitative evaluation of intravenous iron formulations,to provide a reference for drug selection in medical institutions and rational clinical drug use. Methods Based on the A Quick Guideline for Drug Evaluation and Selection in Chinese Medical Institutions (the 2nd edition),the quantitative evaluation of four intravenous iron formulations,including four originator drugs and five generic drugs were conducted across five dimensions: pharmaceutical properties, effectiveness,safety, economy,and others. Results Except for iron dextran generic drugs, all originator drugs and generic drugs of intravenous iron formulations included in the evaluation achieved an overal score of over 70 points. Among them,ferric carboxymaltose (Ferinject) obtained the highest overall score (79.45 points),demonstrating advantages in pharmaceutical characteristics,effectiveness and safety. In the dimension-specific comparison, iron sucrose (Aiyishu) ranked first in the economic dimension (10 points), while iron dextran (CosmoFer) topped the list in other atribute dimensions (8 points). Conclusion Except for iron dextran generic drugs, all evaluated originator drugs and generic drugs of intravenous iron formulations scored above 70 points, indicating that they can allbe strongly recommended for intravenous iron supplementation. Among them, feric carboxymaltose (Ferinject) is the preferred option. Based on the comprehensive evaluation results of intravenous iron formulations derived from the Guideline,this study provides a scientific basis for evaluating and selecting intravenous iron formulations,as well as their rational use in clinical practice.
【Keywords】 Intravenous iron formulations; Iron sucrose; Iron dextran; Ferric derisomaltose; Ferriccarboxymaltose; Drug selection; Drug evaluation
静脉铁剂是一类通过静脉注射或输注给药的铁补充剂,主要用于治疗铁缺乏或缺铁性贫血。(剩余14233字)